In 1900, Moses Gomberg, Professor of Chemistry at the University of Michigan, confirmed the existence of a stable, trivalent organic free radical: triphenylmethyl. In so doing, he challenged the then prevailing belief that carbon could have only four chemical bonds. Gomberg’s discovery made a major contribution to theoretical organic chemistry and fostered a field of research that continues to grow and expand. Today, organic free radicals are widely used in plastics and rubber manufacture, as well as medicine, agriculture and biochemistry.
Organic Chemistry


Havemeyer Hall was built between 1896 and 1898 under the leadership of Charles Frederick Chandler. It provided research and teaching facilities for faculty and students specializing in industrial, inorganic, organic, physical, and biological chemistry. Pioneering research done here led to the discovery of deuterium, for which Harold Clayton Urey received the Nobel Prize in 1934. Six others who did research here subsequently received the Nobel Prize, including Irving Langmuir, the first industrial chemist to be so honored, in 1932.
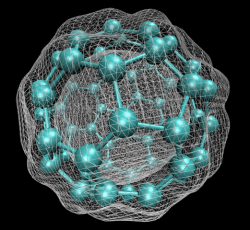
In early September 1985, a team of scientists discovered a previously unknown pure carbon molecule, C60, which they dubbed buckminsterfullerene. The name was chosen because the geodesic domes of Buckminster Fuller provided a clue that the molecule’s atoms might be arranged in the form of a hollow cage. The structure, a truncated icosahedron with 32 faces, 12 pentagonal and 20 hexagonal, has the shape of a soccer ball.
Innovations
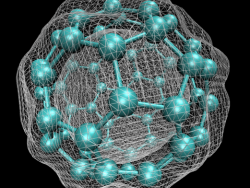
In early September 1985, a team of scientists discovered a previously unknown pure carbon molecule, C60, which they dubbed buckminsterfullerene. The name was chosen because the geodesic domes of Buckminster Fuller provided a clue that the molecule’s atoms might be arranged in the form of a…
Read More
Havemeyer Hall was built between 1896 and 1898 under the leadership of Charles Frederick Chandler. It provided research and teaching facilities for faculty and students specializing in industrial, inorganic, organic, physical, and biological chemistry. Pioneering research done here led to the…
Read More
In 1900, Moses Gomberg, Professor of Chemistry at the University of Michigan, confirmed the existence of a stable, trivalent organic free radical: triphenylmethyl. In so doing, he challenged the then prevailing belief that carbon could have only four chemical bonds. Gomberg’s discovery made a…
Read More

